WP 06: Training
Objectives
The main objective is to help ensure successful performance of the participating sites in terms of state of the art delivery of therapeutic hypothermia, standardised stroke care and adherence to Good Clinical Practice (GCP).
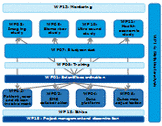
Work package Description
WP6 is be led by Dr. Jesper Petersson (SUS) and Prof. Hanne Christensen (RegionH).
Task 1: Develop comprehensive Clinical Instructions (CI’s). SOP’s will define the areas to be covered by CI’s. CI’s will be based on the study protocol ESO guidelines for treatment of acute ischaemic stroke, and international GCP standards. CI’s will be translated into other languages than English based on National Coordinators wish.
(1) Patient inclusion
(2) Delivery of hypothermia
(3) Monitoring, maintaining homeostasis and prevention of stroke-related complications: This CI will be based on the ESO guidelines on treatment of acute ischemic stroke; standard evidence based stroke care procedures including swallowing test, prevention of deep venous thrombosis, urinary retention, constipation and falls will be included.
(4) Patient follow up and outcome adjudication
Task 2 Define standard criteria for the Certification of Therapeutic Cooling Stroke Units based on current literature and the experience of the investigators
Task 3: E-learning and test series reflecting the procedures of each of the five CI’s will be set up. Further, e-learning in Good Clinical Practice (GCP), National Institute of Health Stroke Scale (NIHSS) and modified Rankin Score (mRS) will be provided. The e-learning series will be prepared in English and translated into other languages based on the National Coordinator’s discretion. E-learning and testing for certification will be set up in the same test and accessible at the study web-site. Investigators will undergo certification.
Task 4: Face-to-face training for investigators
Task 5: Coaching program. In order to harmonise study intervention and care as well as improving patients’ safety, sites will have access to coaching during the procedure. A group of specially trained health professionals will be accessible for sites 24/7 by a hotline telephone. When randomisation of a patient occurs, a text-message to the coach will automatically be generated by the study web-site. The coach will then be able to guide the site through the induction of therapeutic hypothermia or the set up of monitoring in control patients, respectively. The centre will be able to contact the coach at any time. The coaches will have a contact-list of designated experts at regional or local study centres in case further advice is needed. After a completed treatment the coaching-team will review the procedure with site-staff by telephone conference using a structured feedback interview. A continuously updated FAQ (Frequently Asked Questions) database covering questions and answers accessible on-line at the study website will be developed by the coaching team.
WP Leader
Leader: ![]() Dr. Jesper Petersson (SUS)
Dr. Jesper Petersson (SUS)
Co-leader: ![]() Prof. Hanne Christensen (RegionH)
Prof. Hanne Christensen (RegionH)
Participating Partners
Medical University of Vienna
Max Planck Society for the Advancement of Science
Technical University of Denmark
Mayo Clinic Rochester
The University Medical Center Groningen
University of Cambridge
EMC microcollections
Hycult biotechnology